Introduction
The motion of fluids is complex enough when we only consider one “phase” of matter at a time, i.e. a purely liquid or purely gaseous flow. However many problems of practical interest involve liquids and gases simultaneously. This is what we call “multiphase flow”, and it is famously difficult both to measure experimentally and simulate numerically.
Of particular interest in multiphase flows is when we have complex interactions between the two phases. An example of this is “atomization”, where a continuous liquid region breaks up into a cloud of droplets. The spray from a garden hose or a deoderant can are examples of atomization processes. There are actually two stages of atomization, primary and secondary.
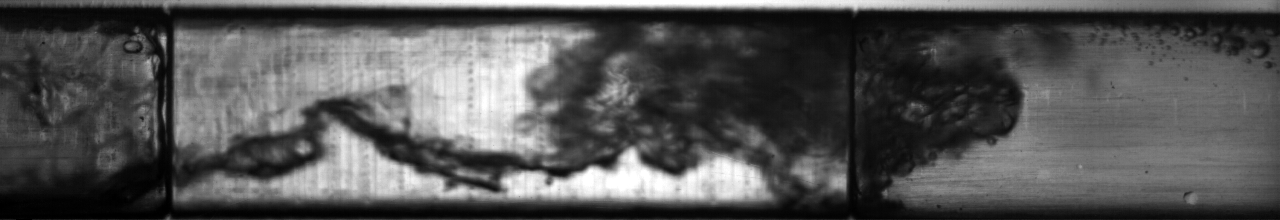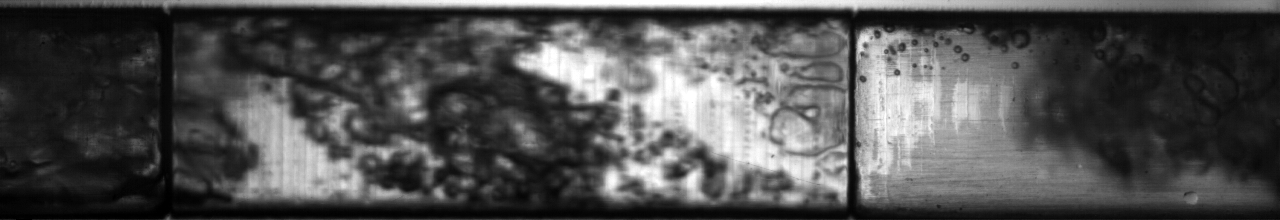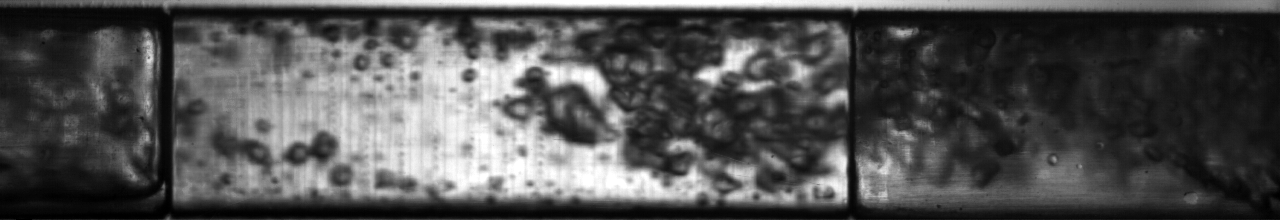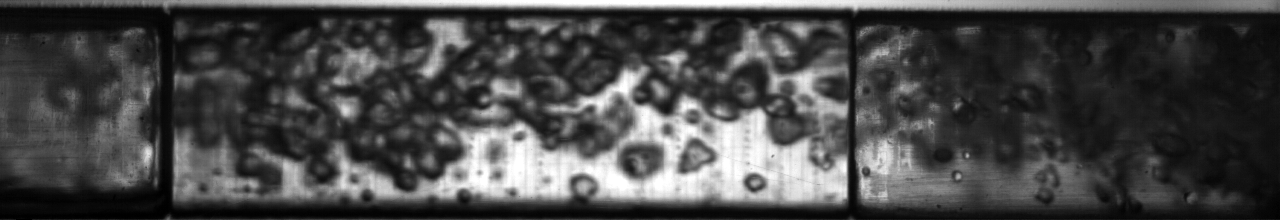
Multiphase flow also produces interesting phenomena when we have fluids of very different density moving past each other; think of the wind blowing across the surface of water and the complex waves that result from it. An example of this situation in two-phase flow of refrigerant can be seen below:

Primary Atomization:
Primary atomization is where a continuous liquid region, like a jet of water from a nozzle, breaks up into a cloud of large droplets. This is beautifully visualized in the work of my colleague, Dr. Daniel Duke (presently at Argonne National Laboratories), in the video below.
Secondary Atomization:
If the droplets are sufficiently large, and the motion of the gas sufficiently violent, the droplets may breakup further, in a process called secondary atomization. In the animation below, a single fluid droplet breaks up in a “bag and stamen” mode. There are a number of different forms this secondary breakup may take, check out the Gallery page for more examples.

Cavitation:
Another multi-phase flow phenomenon that interests us is cavitation. Cavitation occurs in flowing liquids, when something about the flow causes a localized drop in pressure. When the pressure drops, so does the temperature required to boil the liquid – if the local pressure drop is strong enough, this can result in localized boiling, even in an otherwise cold liquid! Cavitation plays a key role in fuel spray processes, as well as having the potential to cause damage to ships propellers and in pipes.
See below for an example of R-134A (the propellant used in asthma puffers, as well as in many refrigerant systems) cavitating while flowing through a transparent 3-D printed part.